
4D-310 is administered via IV delivery and is designed to produce both high, stable AGA activity in the bloodstream and to generate AGA activity intracellularly within critical affected organs, including the heart.ĤDMT is a clinical-stage gene therapy company pioneering the development of product candidates using targeted and evolved AAV vectors. Progression of the disease causes significant reduction in the quality of life and significant economic burden associated with greater patient needs for supportive care.īy using a targeted and evolved vector to deliver a functional copy of the GLA gene, we believe 4D-310 has the potential to be a promising treatment of Fabry disease.
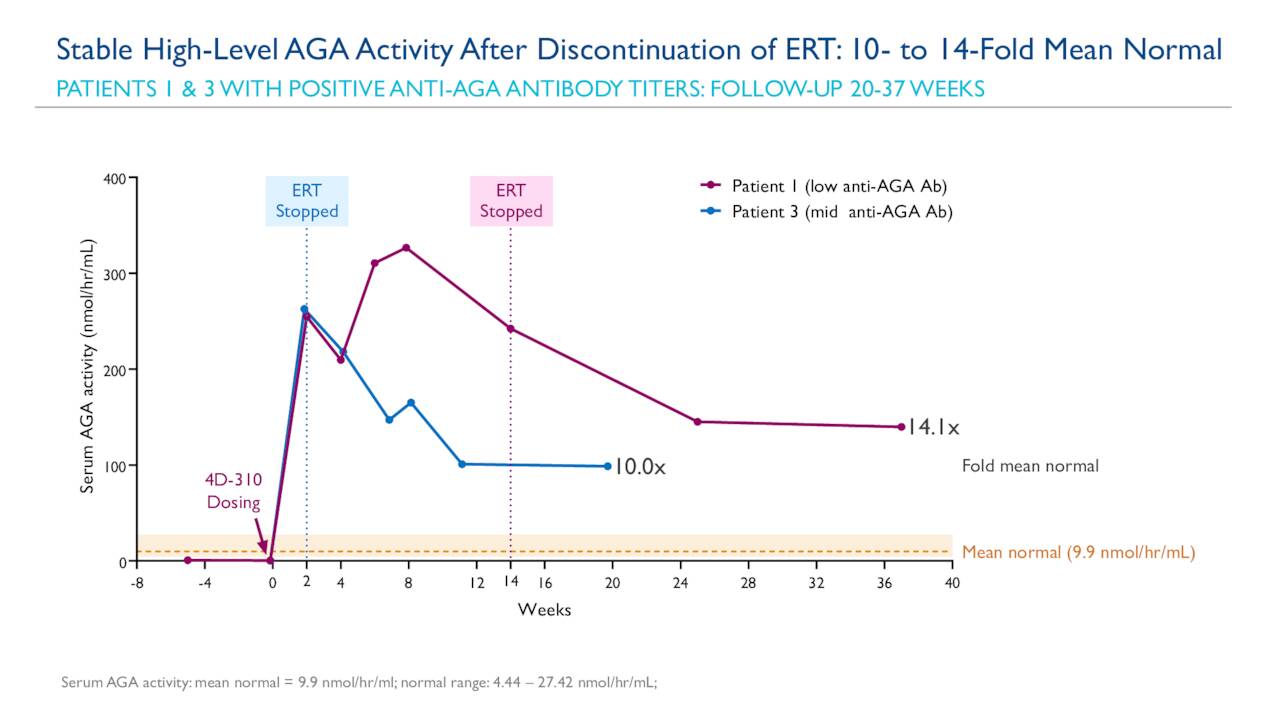
Such substrate accumulation can lead to life-threatening hypertrophic cardiomyopathy, heart failure, arrhythmias, various degrees of kidney dysfunction and cerebrovascular stroke. Jerry Vockley MD, PhD, chief of genomic and genetic medicine.Īffecting more than 50,000 people in the United States and European Union, Fabry disease is a genetic disorder of the GLA gene that results in the body’s inability to produce an enzyme called alpha-galactosidase or AGA, causing the accumulation of the substrate globotriaosylceramide-3 (Gb3) in critical organs, including the heart, kidney and blood vessels. The first patient was dosed at University of Pittsburgh Medical Center (UPMC), Children’s Hospital of Pittsburgh under the direction of principal investigator Dr. Key secondary endpoints include the assessment of biological activity including AGA enzyme activity and substrate concentrations in the blood over time. The study is designed to assess the preliminary safety, tolerability, and maximum-tolerated or maximum-feasible dose of 4D-310. The Phase 1/2 open-label, dose-exploration and dose-expansion study is expected to enroll up to 18 Fabry disease patients. “The targeted organ distribution of 4D-310 in Fabry disease makes it likely that this therapy can also potentially treat the cardiomyopathy associated with Fabry disease, a critical disease manifestation that other therapies have been unable to address.” Raphael Schiffmann, MD, Senior Vice President & Therapeutic Area Head, Cardiology at 4DMT. Gene therapy represents a promising therapeutic modality for patients with Fabry disease because of its potential as a one-time therapy that can deliver stable and sustained levels of AGA activity,” said Dr. Cardiovascular disease is still the leading cause of death in patients despite standard therapy. Current standard of care enzyme replacement therapies are associated with a high-treatment burden, and due to the short half-life in the blood, patients lack therapeutic concentrations of AGA between infusions. “The unmet need for patients with Fabry disease is significant.
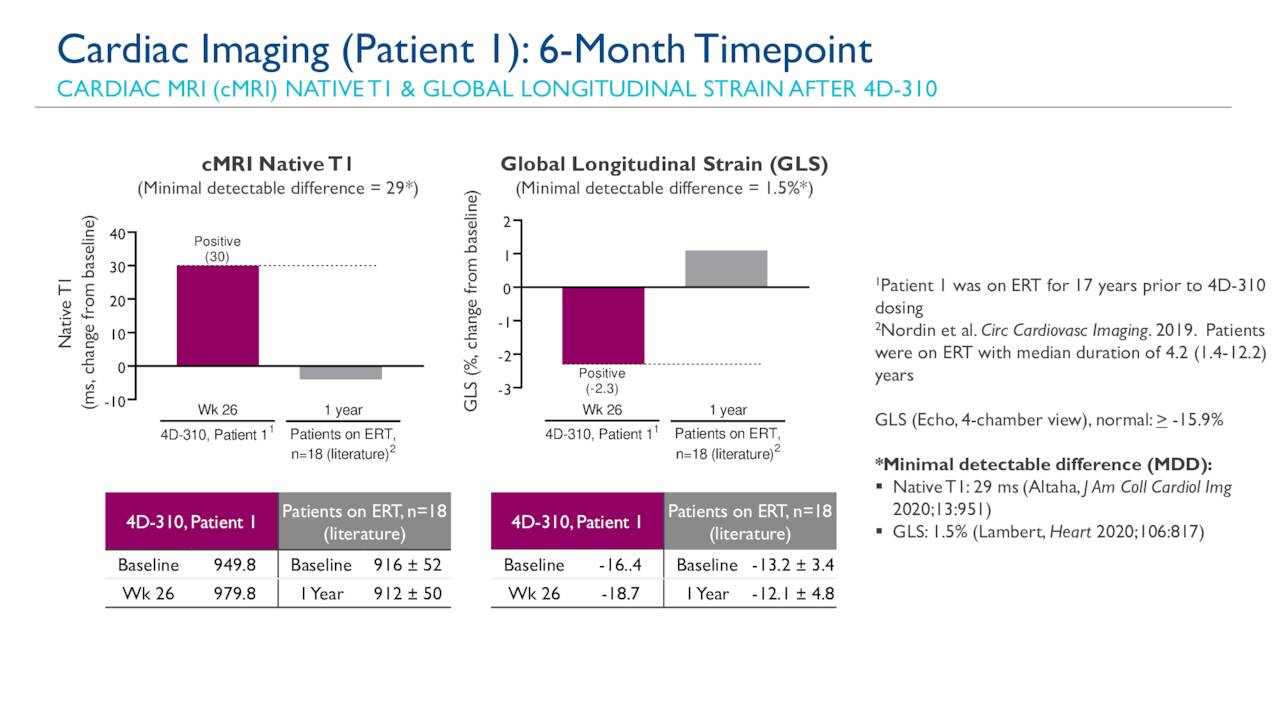
4D-310 is designed for both stable therapeutic AGA enzyme activity in the blood, as well as for intracellular production within affected tissues, including in cardiac muscle cells.” “4D-310 is designed with the goal of achieving a novel dual mechanism-of-action for these patients.
#Emeryville ca 4d molecular therapeutics trial
Dosing the first patient in the Phase 1/2 clinical trial of 4D-310 marks the third product candidate to be administered to patients, all of which utilize proprietary vectors derived from our Therapeutic Vector Evolution platform,” said David Kirn, MD, chief executive officer, co-founder and president of 4DMT. “Through our Therapeutic Vector Evolution platform we apply the principles of directed evolution to invent targeted and evolved AAV vectors for the delivery of genes to specific tissue types. Fabry disease is an inherited lysosomal storage disease with high unmet medical need that results from loss of function mutations in the alpha-galactosidase (AGA) enzyme. EMERYVILLE, CA, USA I MaI 4D Molecular Therapeutics (Nasdaq: 4DMT), a clinical-stage gene therapy company harnessing the power of directed evolution for targeted gene therapies, announced that the first patient has been dosed in the Phase 1/2 clinical trial of 4D-310 for the treatment of Fabry disease.


 0 kommentar(er)
0 kommentar(er)
